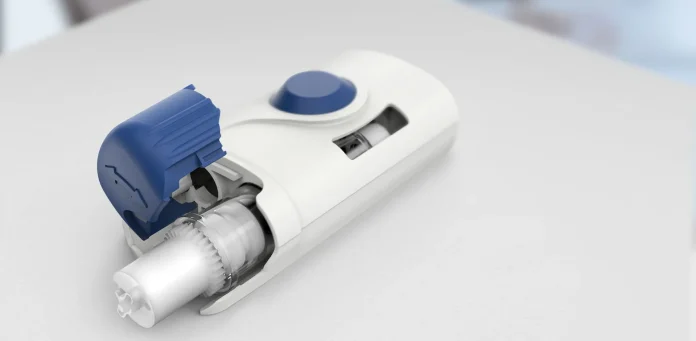
The Wearable Injectors Market is advancing into a new stage of clinical validation and pharmaceutical collaboration. On July 23, 2025, BD (Becton, Dickinson and Company) announced the first pharma-sponsored combination product clinical trial using the BD Libertas Wearable Injector for subcutaneous delivery of complex biologics. This development signals growing pharmaceutical confidence in wearable drug delivery platforms designed to enhance patient convenience and therapeutic flexibility. The transition from internally led studies to pharma-sponsored trials represents a meaningful step in commercial validation.
BD Libertas Wearable Injector and Market Momentum
The selection of BD Libertas for a pharma-sponsored trial follows successful outcomes from more than 50 BD-conducted pre-clinical and clinical studies. According to the announcement, a device clinical study demonstrated excellent performance, with 100% of study participants stating they would likely use the BD Libertas Wearable Injector if prescribed. This level of patient-reported acceptance is significant in the wearable injectors market because usability and comfort directly influence real-world adoption. The pharma-sponsored combination product clinical trial is described as an advancement in accelerating innovation in drug-device combination products. It also supports potential conversion from infused medications that require hospital or clinic visits to more convenient self-injection in various care settings, including at home.
Wearable Injectors Market and the Biologics Expansion
The biologics market remains a primary driver for wearable injector innovation. According to the announcement, the biologics market is expected to grow to more than $670 billion by 2030. Biologic therapies often require delivery of high-viscosity and large-volume formulations. The BD Libertas Wearable Injector supports delivery of biologics up to 50 centipoise and is offered in 2 to 5 mL and 5 to 10 mL configurations. The system features a fully mechanical, patient-ready-to-use design with a “peel, stick and click” mechanism and does not require end-user filling or assembly. These features directly address operational and usability challenges associated with traditional infusion-based biologic therapies.
Clinical Evidence Supporting Market Confidence
Clinical validation plays a central role in strengthening confidence in the wearable injectors market. The referenced early feasibility clinical study evaluated 5 mL subcutaneous placebo injections in 52 healthy adult subjects. According to the announcement, peak pain measured on a 100 mm Visual Analog Scale averaged 9.1 mm and rapidly resolved within 30 minutes, with a mean of 0.4 mm. Where tissue effects were observed, the majority resolved within 60 minutes and all resolved within 24 hours. Additionally, 100% of participants stated that they would likely use the device if prescribed. These findings demonstrate tolerability, functionality, and patient acceptability, all of which are essential for drug-device combination products.
Manufacturing Readiness and Commercial Scalability
Beyond clinical validation, manufacturing scalability is critical for sustained growth in the wearable injectors market. BD stated that it is conducting ongoing validations of fill-finish and final assembly processes with multiple Contract Manufacturing Organizations. This enables support for pharmaceutical partners from development through commercial-scale production. This infrastructure reduces operational risk and supports integration into pharmaceutical pipelines. It also strengthens the commercialization pathway for drug-device combination products.
Wearable Injectors Market: BD Libertas Key Highlights
| Category | Key Detail | Impact |
| Clinical Trial | First pharma-sponsored combination product trial announced July 2025 | Signals pharma validation |
| Patient Acceptance | 100% participants likely to use if prescribed | Strong usability confidence |
| Delivery Capability | Supports up to 50 cP; 2–10 mL configurations | Enables large-volume biologic delivery |
| Biologics Outlook | Market expected to exceed $670 billion by 2030 | Expands long-term growth opportunity |
Next Steps
- Pharmaceutical companies should evaluate opportunities to integrate wearable injectors into biologic development pipelines, particularly for therapies currently dependent on infusion-based administration.
- Medical technology developers should continue investing in large-volume and high-viscosity delivery capabilities to align with biologic formulation requirements.
- Healthcare providers should assess the operational and patient-experience benefits associated with transitioning eligible therapies to subcutaneous wearable delivery.
- Contract Manufacturing Organizations should prioritize validated fill-finish and final assembly processes to support combination product scalability.
- Investors and industry stakeholders should monitor ongoing clinical milestones and pharma-device collaborations as leading indicators of wearable injectors market expansion.
Conclusion
The first pharma-sponsored clinical trial using BD Libertas Wearable Injector represents a defining moment in the wearable injectors market. It reflects increased pharmaceutical trust, validated patient acceptance, and alignment with the rapidly expanding biologics sector. With strong clinical data, scalable manufacturing validation, and support for home-based administration, wearable injector technologies are positioning themselves as a critical component of next-generation biologic therapy delivery. The market is no longer driven solely by technological innovation. It is increasingly shaped by collaborative clinical execution and patient-centric design.
About The Author
Borna Dhar is an aspiring content writer with a strong interest in digital transformation, emerging technologies, and industry-focused research. She is building hands-on experience in creating clear, research-driven content that strengthens digital visibility and supports the evolving needs of modern businesses. With exposure to diverse sectors such as technology and digital services, she brings a fresh analytical perspective and contributes to communicating meaningful insights, innovation, and value propositions for niche and targeted audiences.